Selective electrocatalytic hydroboration of aryl alkenes
Time:2023-04-27 14:08 Author:Yahui Zhang
Ce Bi, Xiangyu Zhao, Zhiqi Jia, Yongxin Chang, Hang Yang, Mengyuan Song, Xin Zhang, Yahui Zhang, and Guangyan Qing*
Green Chem. 2021, 23, 1691.
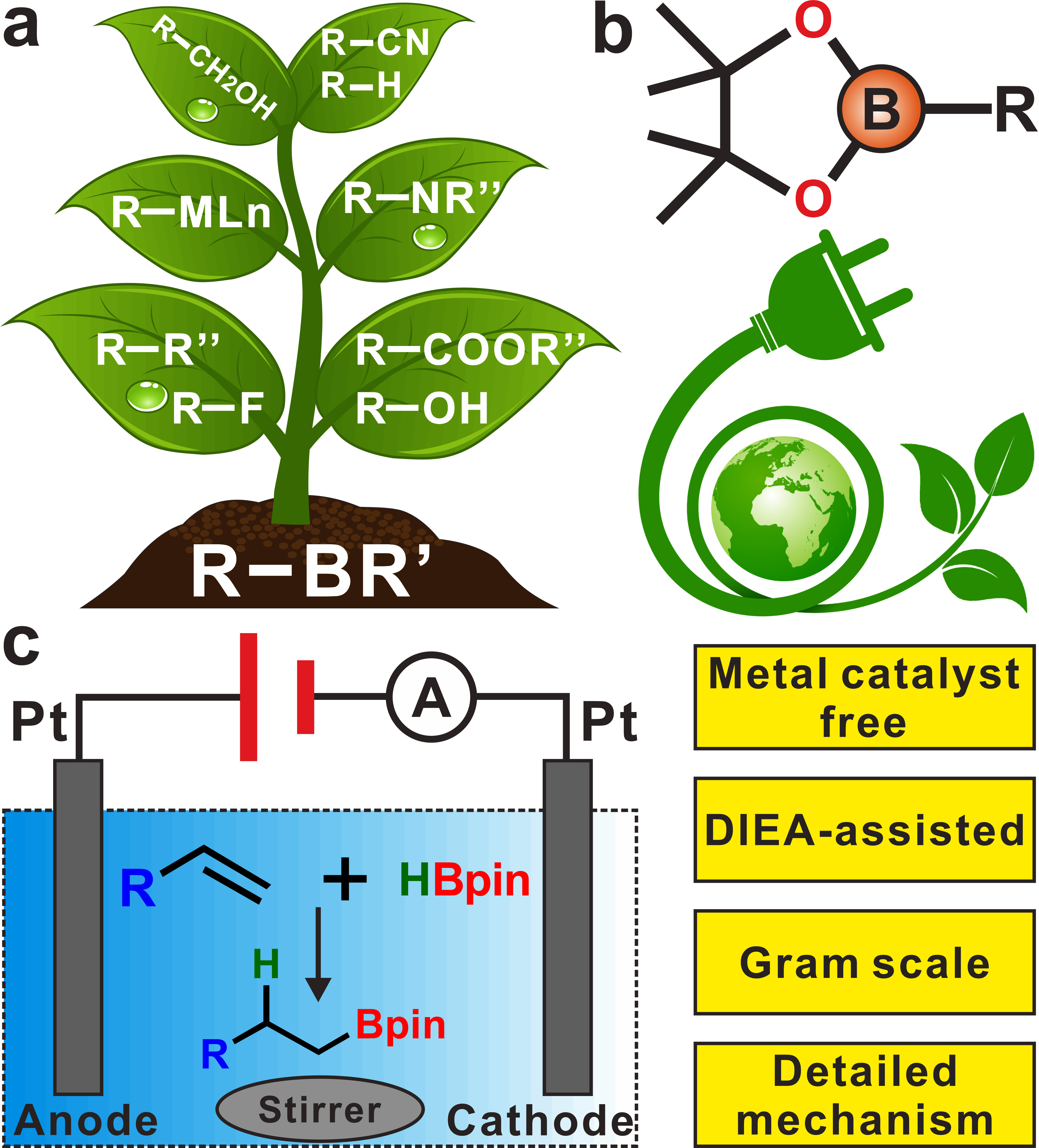
Organoboron compounds are powerful precursors of value-added organic compounds in synthetic chemistry, and transition metal-catalysed borylation has always been dominant. To avoid toxic reagents and costs associated with metal catalysts, simpler, more economical and effective approaches for delivering organoborons are highly desirable. Here, without the use of any metal catalysts, a CH3CN-involved electrochemical borylation reaction is reported with alkenes and HBpin as substrates. The site-selectivity is realised to achieve mono- or di-functional borylation of an unsaturated bond by regulating the proportion of HBpin. In addition, the success of gram-scale experiments and versatile conversions confirms the potential applications of this strategy in industrial synthesis. The vital auxiliary role of N,N-diisopropylethylamine (DIEA) in the process of acetonitrile electrolysis is disclosed in the mechanism study. The proposed new strategy provides a generic platform for the discovery of additional challenging electrochemical systems for hydrogenation reactions.